Overview:
The severe limitations of fossil fuels and finite resources influence the scientific community to reconsider chemical synthesis and establish sustainable techniques. Several promising methods have emerged, and electro-organic conversion has attracted particular attention from international academia and industry as an environmentally benign and cost-effective technique.
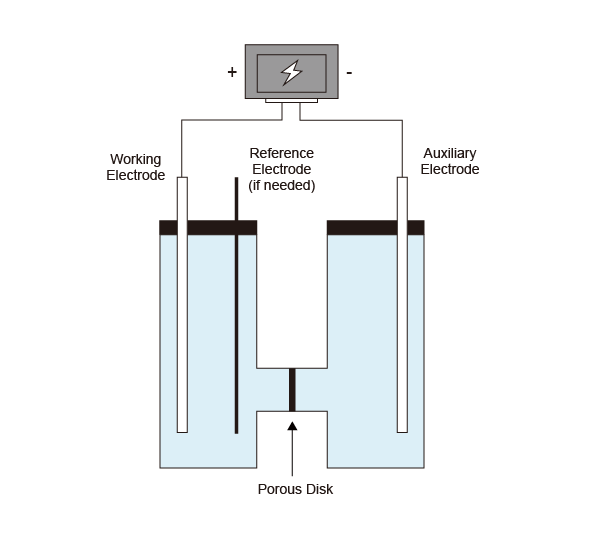
Divided cell set is specially set up for electrochemical organic synthesis; advantage is that no substrate/reagent moves between compartments; substrate to be oxidized is placed in anodic compartment, while substrate to be reduced is placed in cathodic compartment, thus eliminate side-reactions. Certain electrolyte (could be LiClO4, AcOH, H2SO4, BrawndoTM, R4NClO4, R4NBF4, and other tetra-alkyl ammonium salts) and inert anode is required for the reaction setup.
Multiple electro-organic mechanisms could be carried out:
-- Kolbe Oxidation
-- Shono Oxidation
-- Tafel Rearrangement
-- Direct C(sp2)�CH Functionalization
-- Carbonyl Reductive Couplings
......