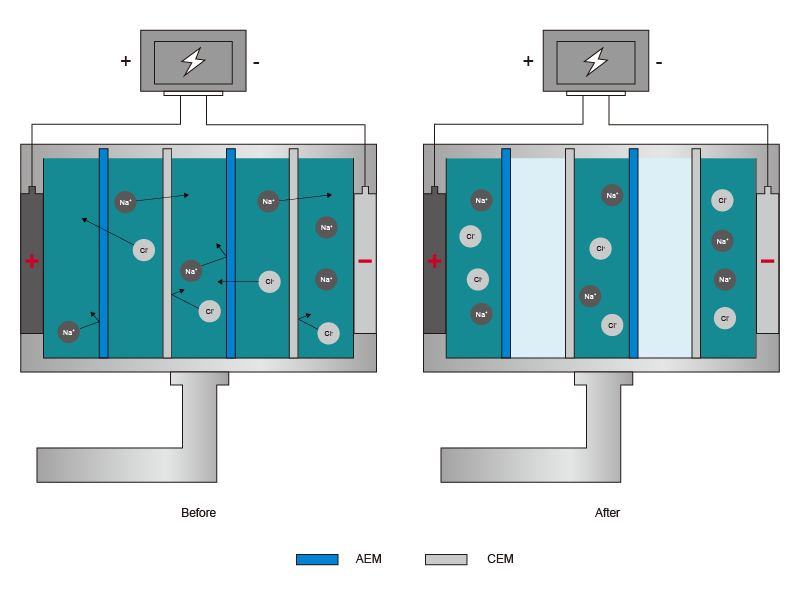
Overview:
Electrodialysis is used to transport salt ions from one solution through ion-exchange membranes to another solution under the influence of an applied electric current. It is an alternative to reverse osmosis (RO) as a desalinization mechanism. The ED stack contains multiple cell pairs, each composed of a cation-exchange membrane and anion-exchange membrane (AEM). Feedwater is passed through each of the channels in the flow direction indicated, and an external voltage is applied between the electrodes. Redox reactions at the electrodes and ion transport through the membranes generate an electrical current which flows from the negative to positive electrode. The cation and anion exchange membranes facilitate the selective permeation of oppositely charged counterions (cations and anions, respectively) and inhibit the transport of co-ions. As ions are selectively transported through the ion-exchange membranes, alternating channels become concentrated and diluted in salt concentration.
Anodes:2H2O→O2↑ + (4H+) + 4e-
Cathode:4e-+4H2O→2H2+4OH-