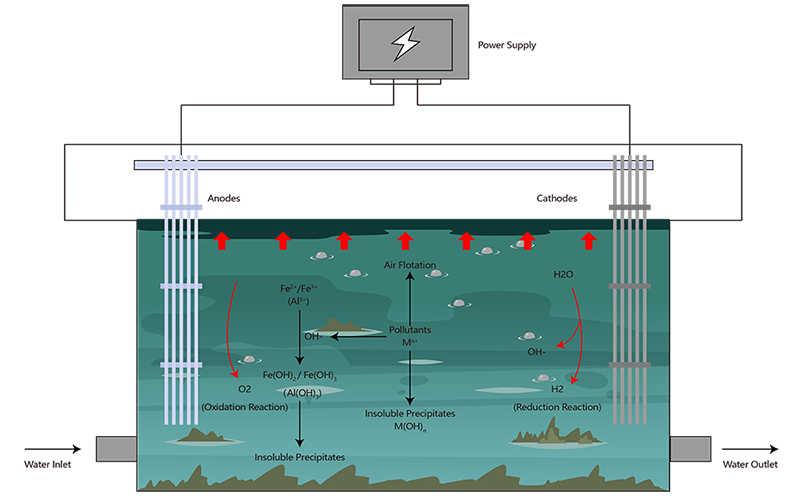
1. Electroflocculation-Electroflotation
When powered by a DC power source, anode material (Aluminum, Iron, etc.) will dissolve into the surrounding environment in the form of cations. When subject to the processes of hydrolysis, polymerization and oxidation, the solution produced is a mixture of hydroxy complex, polynuclear hydroxy complex and hydroxide colloid. Subsequently this will improve agglomeration precipitation of colloidal/suspended impurities.
Any charged pollutant would be attracted by the electric field and neutralized by the electrode, thus destabilizing and precipitating the particles.
Simultaneously the reduction reaction taking place at the cathode side would be constantly producing hydrogen, which absorbs hydrophobic substances and any lightweight particles suspended in the environment whilst floating to the surface. Separation of pollutants from the liquid medium can be achieved using this process and therefore it can be utilized in the purification of sewage.
2. Electrochemical Oxidation
Electrochemical oxidation is an effective method used in the treatment of high-concentration organic wastewater. This process degrades complex organic pollutants, present in wastewater, into non-toxic small-molecule organic compounds or carbon dioxide and water. This is achieved by the mineralization of the various pollutants.
Electrochemical oxidation is divided into 2 forms, direct oxidation and indirect oxidation, both of which are anodic processes. Direct oxidation is the direct conversion of pollutants into harmless substances (CO2 and H2O) through anodic oxidation; Indirect oxidation produces intermediate substances with a strong oxidation effect through the anodic reaction (or other intermediate reactions) to oxidize the treated pollutants and finally convert them into harmless substances for disposal.








